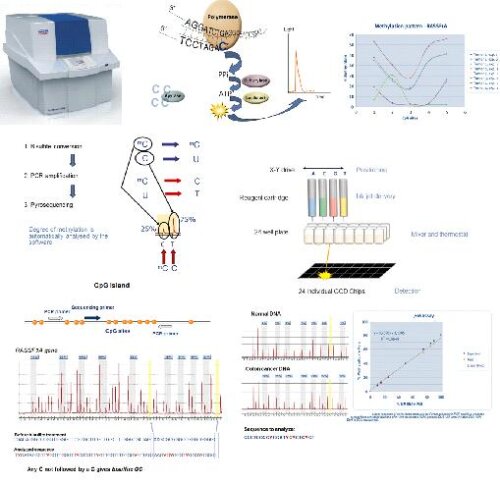
The CDS lab has standard access to two university facilities, allowing for frequent measurement on up-to-date instrumentation.
Pyrosequencing (SNP, CpG)
Bisulfite sequencing is the use of bisulfite treatment of DNA to determine its pattern of methylation. DNA methylation was the first discovered epigenetic mark, and remains the most studied. In animals it predominantly involves the addition of a methyl group to the carbon-5 position of cytosine residues of the dinucleotide CpG, and is implicated in repression of transcriptional activity.Treatment of DNA with bisulfite converts cytosine residues to uracil, but leaves 5-methylcytosine residues unaffected. Thus, bisulfite treatment introduces specific changes in the DNA sequence that depend on the methylation status of individual cytosine residues, yielding single- nucleotide resolution information about the methylation status of a segment of DNA.
Following PCR amplification of the region of interest, CpG pyrosequencing is used to determine the bisulfite-converted sequence of specific CpG sites in the region. The ratio of C-to-T at individual sites can be determined quantitatively based on the amount of C and T incorporation during the sequence extension.
Phospho-peptide Kinome Profiling Platform
https://www.kinases-epigenetics-ppes.com/index.html
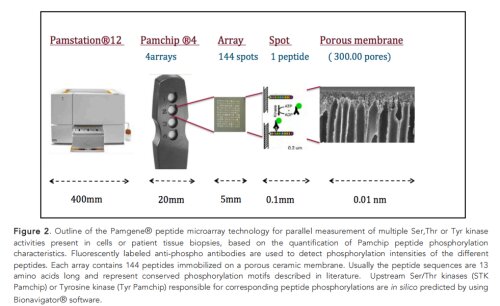
Global kinome activity analysis of kinase signaling pathways using a pamgene-12® peptide array platform
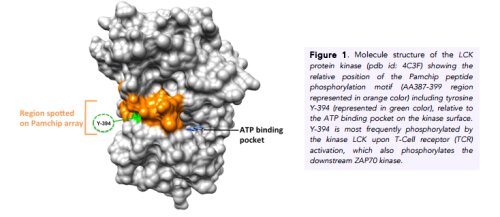
Ser, Thr and Tyr kinases are responsible for the regulation of several cellular processes. When kinases are abnormally expressed or controlled, they promote adverse cancer hallmarks, including cell survival, metastasis and proliferation (1). Reciprocally, protein kinase inhibitor (PKI) drugs have recently joined the arsenal of anti-cancer drugs. In this respect, the fundamental characterization of hyper activated kinases in cancer or kinase inhibition profile following cancer therapy in patients is of utmost importance to optimize therapeutic interventions. Overcoming cancer therapy resistance remains a fundamental challenge for most treatments. Of special note, since the number of FDA approved PKI small molecule drugs has been increased from 1 to 26, peptide array based kinome activity profiling offers new perspectives for patient stratification and tailored cancer therapy design of FDA approved and/or (pre) clinical kinase inhibitors (2). Pamchip® peptide array technology allows parallel measurement of multiple kinase activities in cells as well patient tissues, by profiling peptide phosphorylation characteristics by a pharmacology-on-chip approach (3). Besides its well established anti-cancer applications, Pamchip® peptide array technology also offers innovative opportunities in the field of cardio metabolic health, neurodegenerative disease, as well as pharmacological structure-activity studies of preclinical kinase inhibitors. For more information on scientific publications and brief technology outline visit www.pamgene.com.
Key application areas
Fundamental and discovery research
Target discovery
Pathway identification
Compound mode of action
Biomarker and clinical research
Classification of biomarkers
Therapy predictive biomarkers
Prognostic biomarkers
Technology outline
Readout of microarray data
Data analysis and biological interpretation
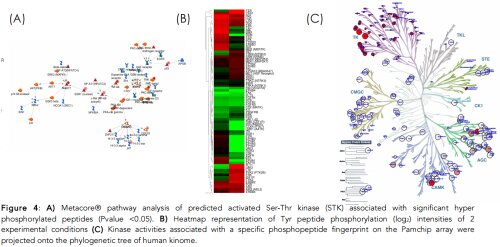
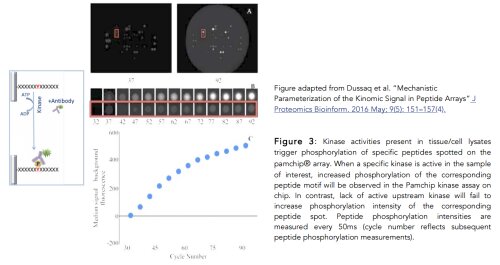
Real time measurements of Pamchip peptide phosphorylation intensities are illustrated in figure 3A and 3B. The median signal-noise background (fluorescence intensity) of each spotted peptide phosphorylation is recorded every 50ms, as summarized in figure 3C. Pamgene Bio-navigator® algorithms based on the phosphonet database (collection of 950,000 known phosphorylation sites in over 21,000 human proteins) allow to convert phosphopeptide intensities into a ranked list of hyper activated/repressed upstream kinases (kinome activity signature) (4). Briefly, fluorescence intensity data of phosphopeptide signals of control and treatment setups are uploaded to Bio-navigator® which implements statistical tests and algorithms for inferring and scoring upstream kinase analysis associated with the Pamchip phosphopeptide profile of interest (Fig. 4B) (5). Outputs of Bionavigator® are compatible with Ingenuity ® or Metacore® pathway analysis, to predict the downstream biological effects or calculation of pathway specific activation or inhibition Z-scores (figure 4A). Finally, the ranked kinome activity signature associated with a Pamchip phosphopeptide profile of interest can be projected onto the phylogenetic kinome tree (figure 4C).
Advantages

References
- Hanahan D, Weinberg RA. Hallmarks of cancer: The next generation. Vol. 144, Cell. 2011. p. 646–74.
- Wu P, Nielsen TE, Clausen MH. FDA-approved small-molecule kinase inhibitors. Vol. 36, Trends in Pharmacological Sciences. 2015. p. 422–39.
- Hilhorst R, Houkes L, Mommersteeg M, Musch J, van den Berg A, Ruijtenbeek R. Peptide microarrays for profiling of serine/threonine kinase activity of recombinant kinases and lysates of cells and tissue samples. Methods Mol Biol. 2013 Jan;977:259– 71.
- Alex Dussaq, 1 Joshua C Anderson, 1 Christopher D Willey 1 and Jonas S Almeida2. Mechanistic Parameterization of the Kinomic Signal in Peptide Arrays. J Proteomics Bioinform. 2016;9(5):151–157.
- Naji F, de Wijn R MA. BioNavigator, software to interpret PamChip® measurements.
UAntwerp core facility for proteomics (CFP-CEPROMA)
Our lab has access to the UAntwerp Core Facility Centre for Proteomics and Mass spectrometry and to the UA-VITO core facility Centre For Proteomics, both of which have state-of-the-art mass spectrometers like ABI 4800 MALDI-TOF-TOF, Micromass QTOF, Waters Synapt Ion mobility QTOF, Waters Xevo triple quadrupole and a Thermo Orbitrap.
This access ensures the possibility of measuring many different aspects from proteins (identification, posttranslational modification, quantification, structure etc.).